NPRA approves shelf-life extensions for several Covid-19 vaccines
This article is a year old
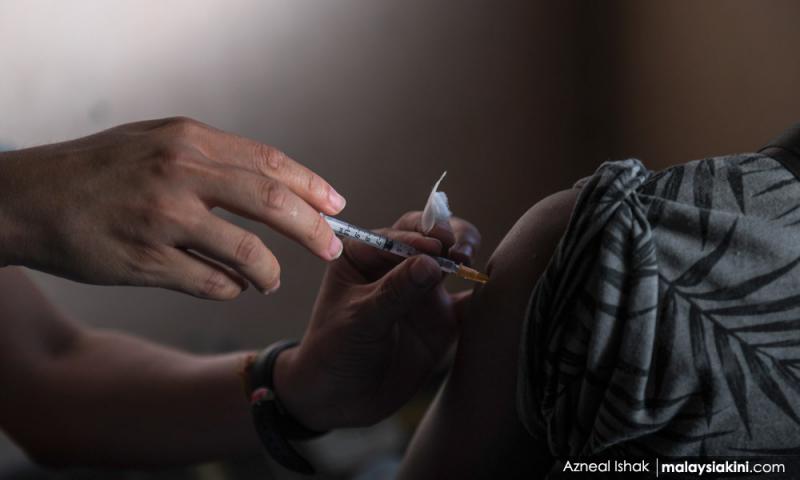
The National Pharmaceutical Regulatory Agency (NPRA) has approved shelf-life extensions for several Covid-19 vaccine products in light of new data submitted by their respective manufacturers.
This gives a new shelf life of 15 to 24 months, depending on the vaccine in question, for the vaccines produced...
Verifying user
RM12.50 / month
- Unlimited access to award-winning journalism
- Comment and share your opinions on all our articles
- Gift interesting stories to your friends
- Tax deductable